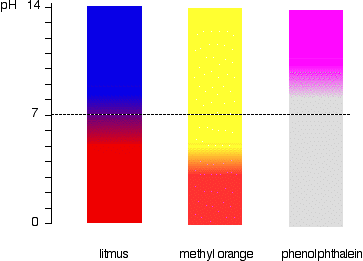
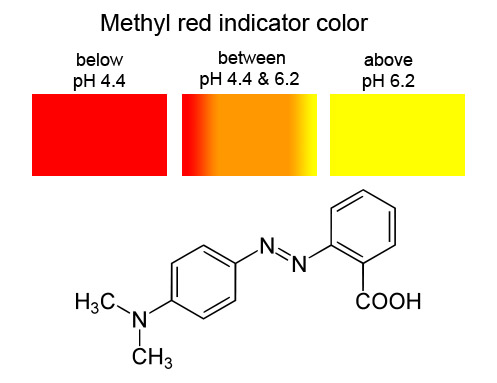
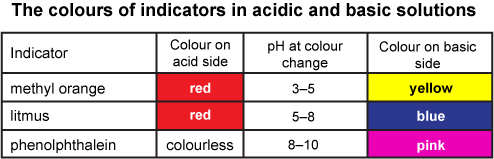
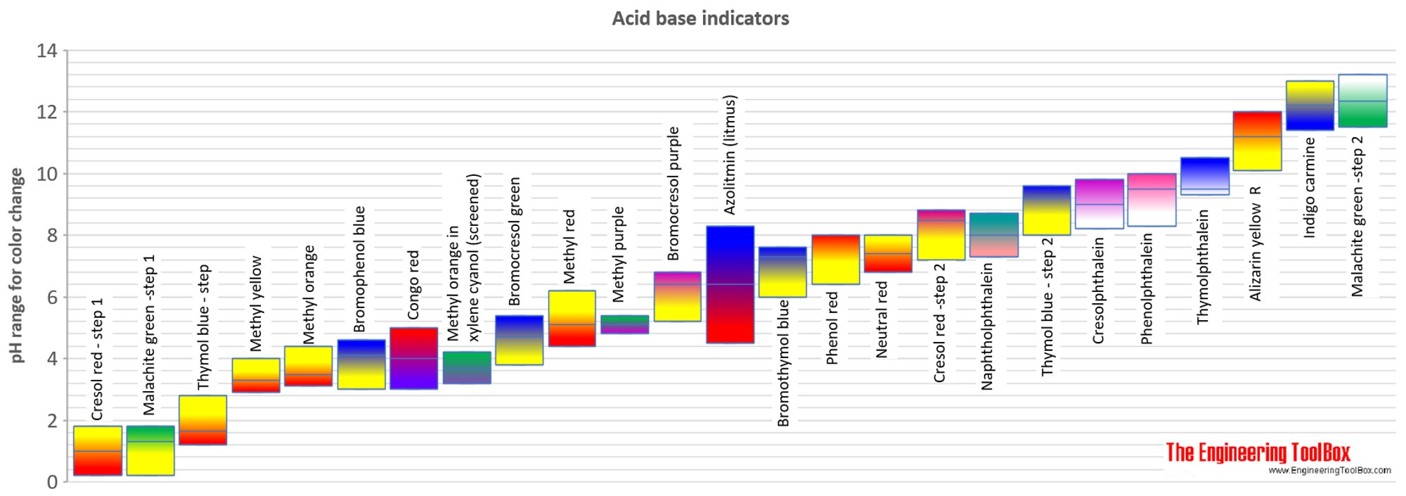
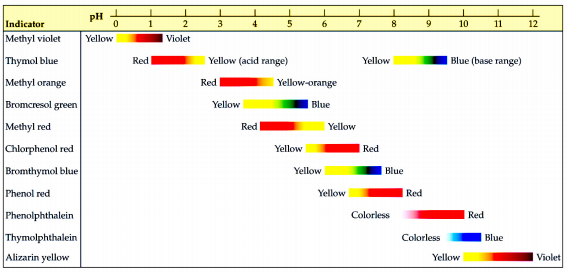
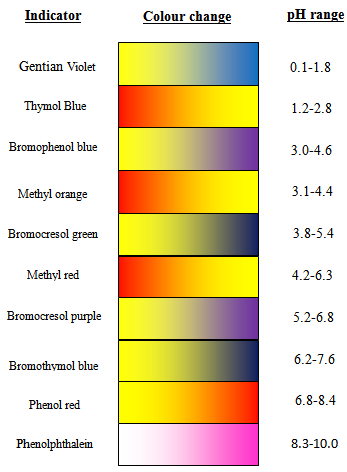
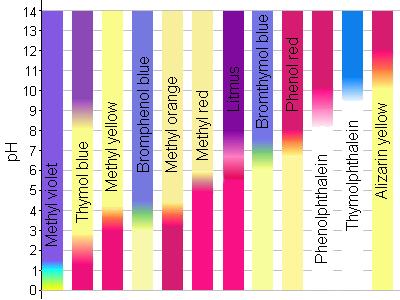
A particular solution produced a yellow colour when tested with both cresol red, methyl orange and bromothymol blue. What does this imply about the pH of the solution? - Quora

uv-visible light absorption spectrum of methyl red and methyl orange in acid or alkaline solution spectra of titration indicators Doc Brown's chemistry revision notes