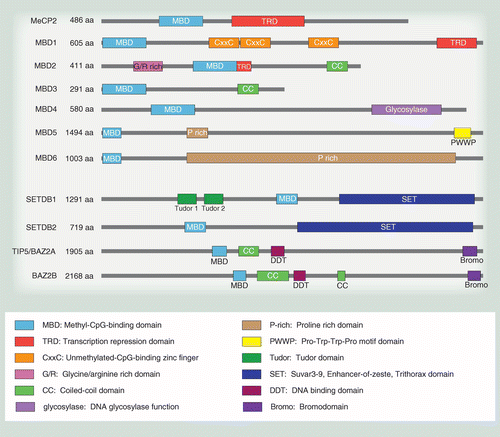
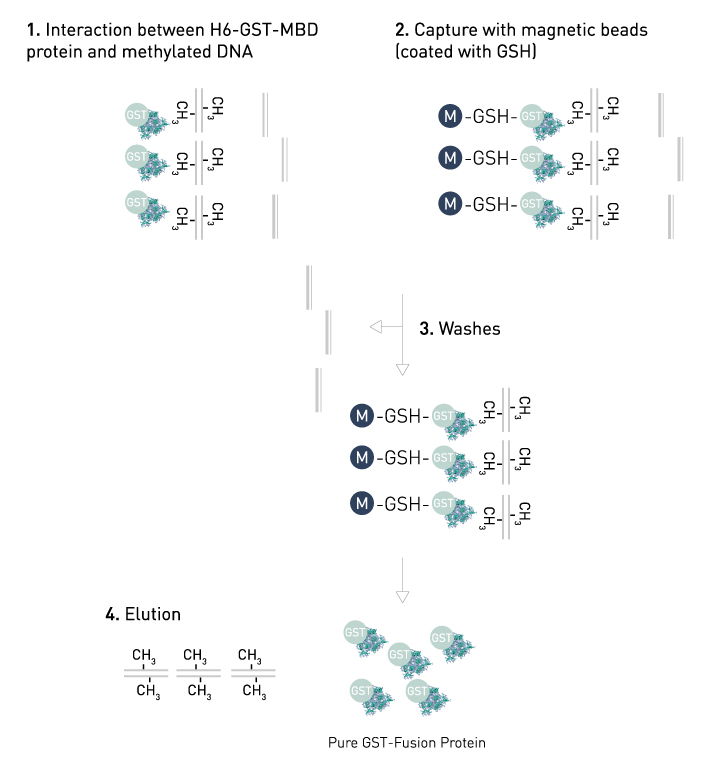
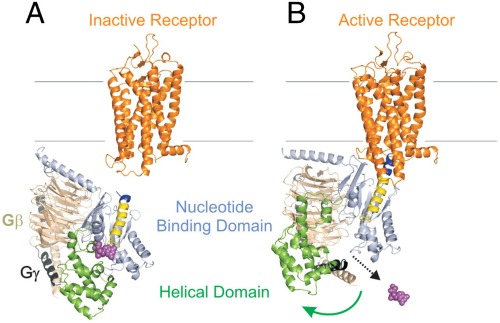
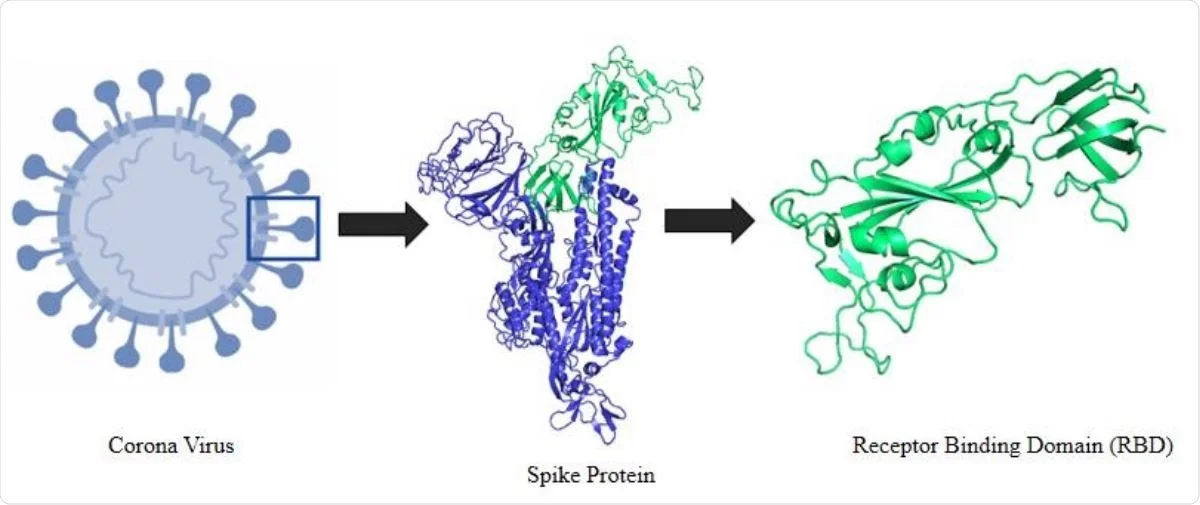
Characterizing the Binding Interactions between DNA-Binding Proteins, XPA and XPE: A Molecular Dynamics Approach | ACS Omega

PDZ domains and their binding partners: structure, specificity, and modification | Cell Communication and Signaling | Full Text
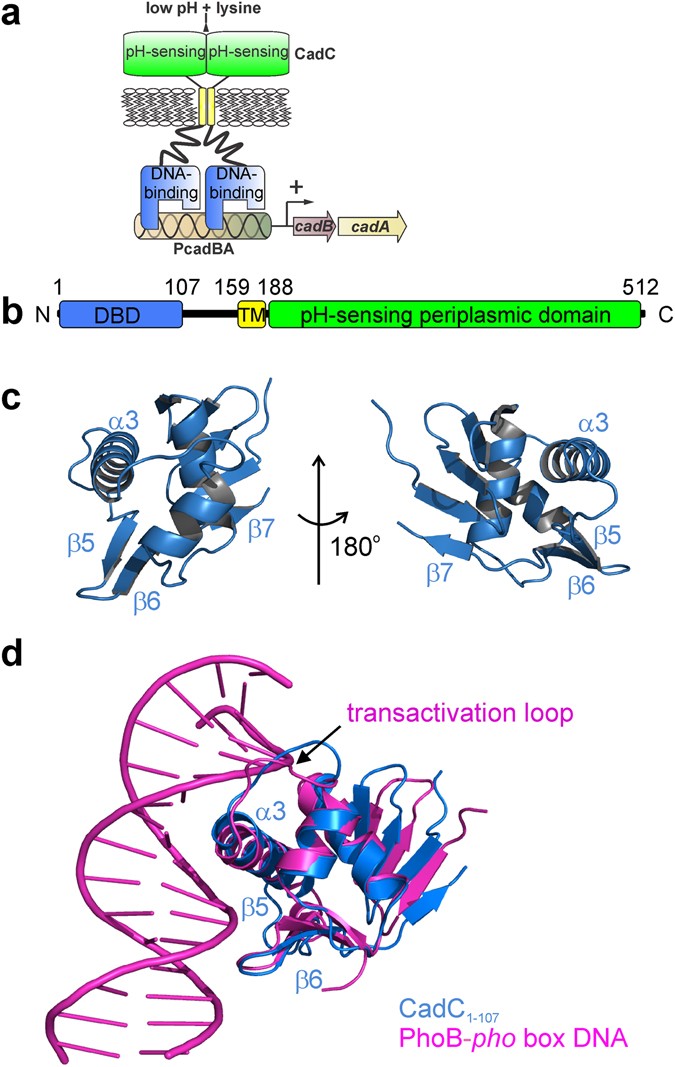
Structure-function analysis of the DNA-binding domain of a transmembrane transcriptional activator | Scientific Reports

A Cross-Reactive Small Protein Binding Domain Provides a Model to Study Off-Tumor CAR-T Cell Toxicity: Molecular Therapy - Oncolytics
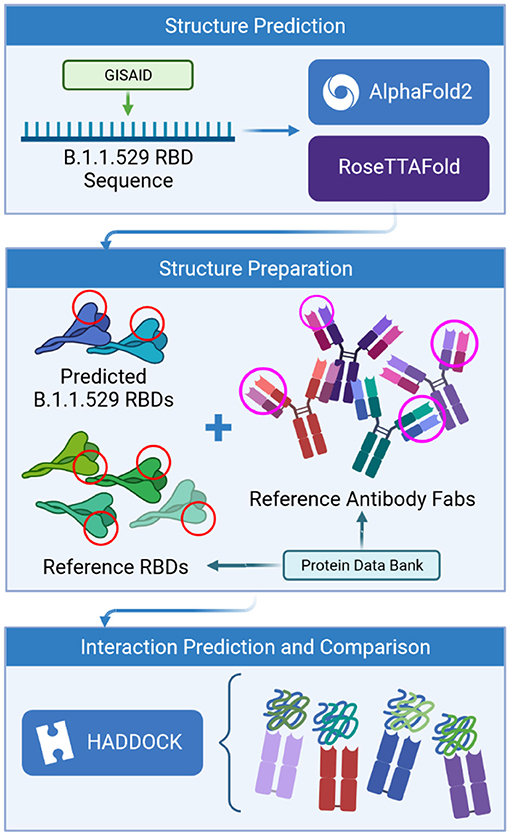
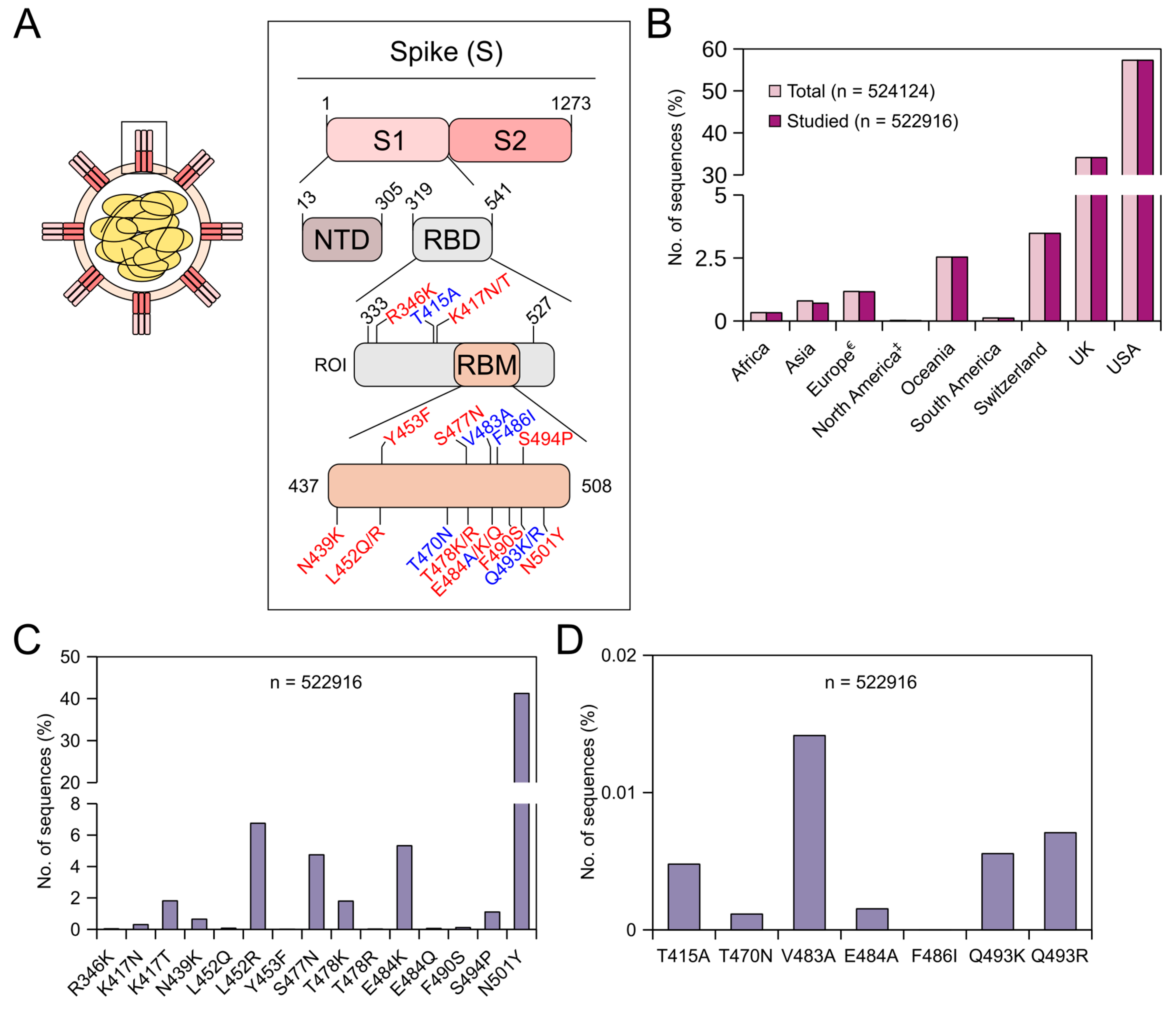
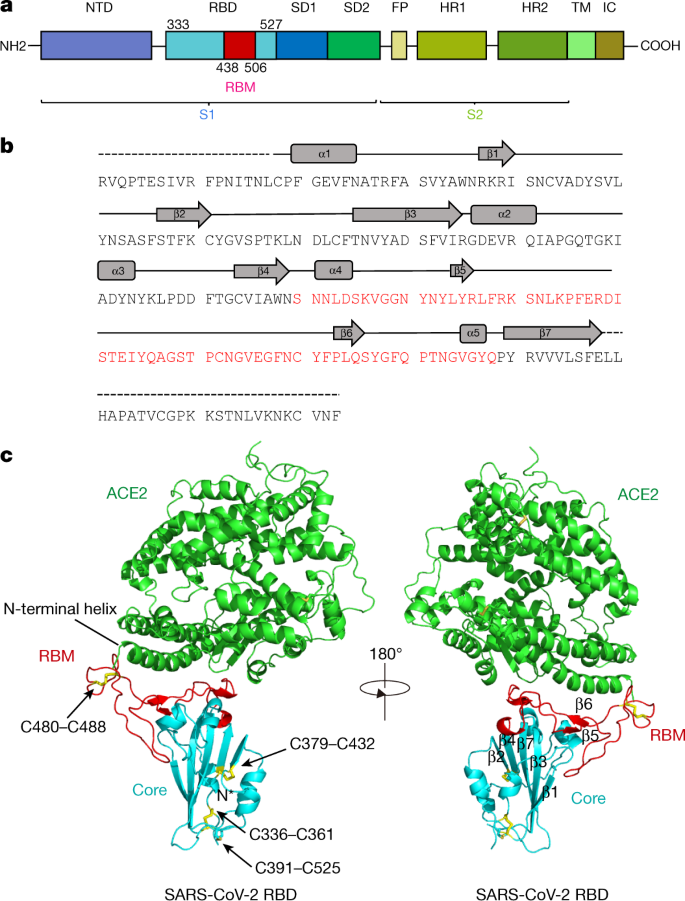
Frontiers | Predictions of the SARS-CoV-2 Omicron Variant (B.1.1.529) Spike Protein Receptor-Binding Domain Structure and Neutralizing Antibody Interactions

Rapid Development of SARS-CoV-2 Spike Protein Receptor-Binding Domain Self-Assembled Nanoparticle Vaccine Candidates | ACS Nano

In Silico Prediction of Compounds Binding to Human Plasma Proteins by QSAR Models - Sun - 2018 - ChemMedChem - Wiley Online Library
![68Ga]ABY-028: an albumin-binding domain (ABD) protein-based imaging tracer for positron emission tomography (PET) studies of altered vascular permeability and predictions of albumin-drug conjugate transport | EJNMMI Research | Full Text 68Ga]ABY-028: an albumin-binding domain (ABD) protein-based imaging tracer for positron emission tomography (PET) studies of altered vascular permeability and predictions of albumin-drug conjugate transport | EJNMMI Research | Full Text](https://media.springernature.com/lw685/springer-static/image/art%3A10.1186%2Fs13550-020-00694-2/MediaObjects/13550_2020_694_Fig1_HTML.png)
68Ga]ABY-028: an albumin-binding domain (ABD) protein-based imaging tracer for positron emission tomography (PET) studies of altered vascular permeability and predictions of albumin-drug conjugate transport | EJNMMI Research | Full Text
Construction and immunogenic studies of a mFc fusion receptor binding domain (RBD) of spike protein as a subunit vaccine against SARS-CoV-2 infection - Chemical Communications (RSC Publishing)
The Modular Organization of Domain Structures: Insights into Protein–Protein Binding | PLOS Computational Biology
Insights into Protein–DNA Interactions through Structure Network Analysis | PLOS Computational Biology

Staphylococcal protein A inhibits complement activation by interfering with IgG hexamer formation | PNAS